Pharma global drug label intelligence is critical in getting products to market and keeping them there, from submission planning to approval and commercialization. As an Authoritative Source, the Dr.Evidence platform empowers you to efficiently and confidently conduct prior precedent and competitive research with automated monitoring, to help inform and de-risk strategy.


Global label intelligence is critical in getting products to market and keeping them there, from submission planning to approval and commercialization. As an Authoritative Source, the Dr.Evidence platform empowers you to efficiently and confidently conduct prior precedent and competitive research with automated monitoring, to help inform and de-risk strategy.

Access approved labels from major geographies globally, including prescription and non-prescription drugs. Leverage contemporary data and streamlined workflows to develop Target Product and Target Label Profiles, support Company Core Data Sheets, and plan for health authority negotiations.

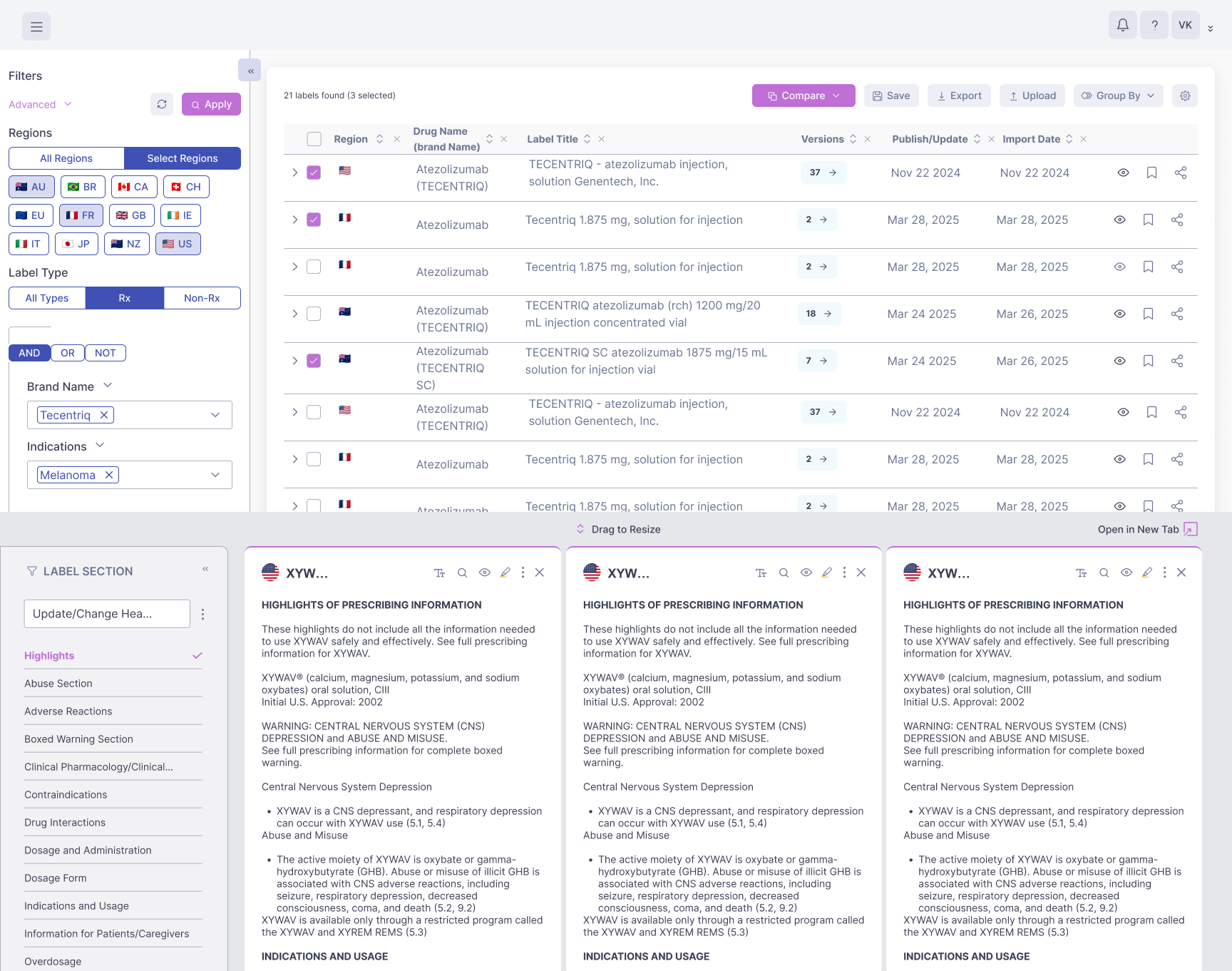
Generate side-by-side comparisons instantly, quickly identify the key differences between label versions, and review adverse events across labels to effectively inform and accelerate label strategy.

Human-in-command AI supports your work, when you need it, by delivering:
With proprietary models trained strictly on labels approved by health authorities and curated by Dr.Evidence, there’s no risk of IP leakage or hallucination. You and your team benefit from precision and recall that outpace industry benchmarks.

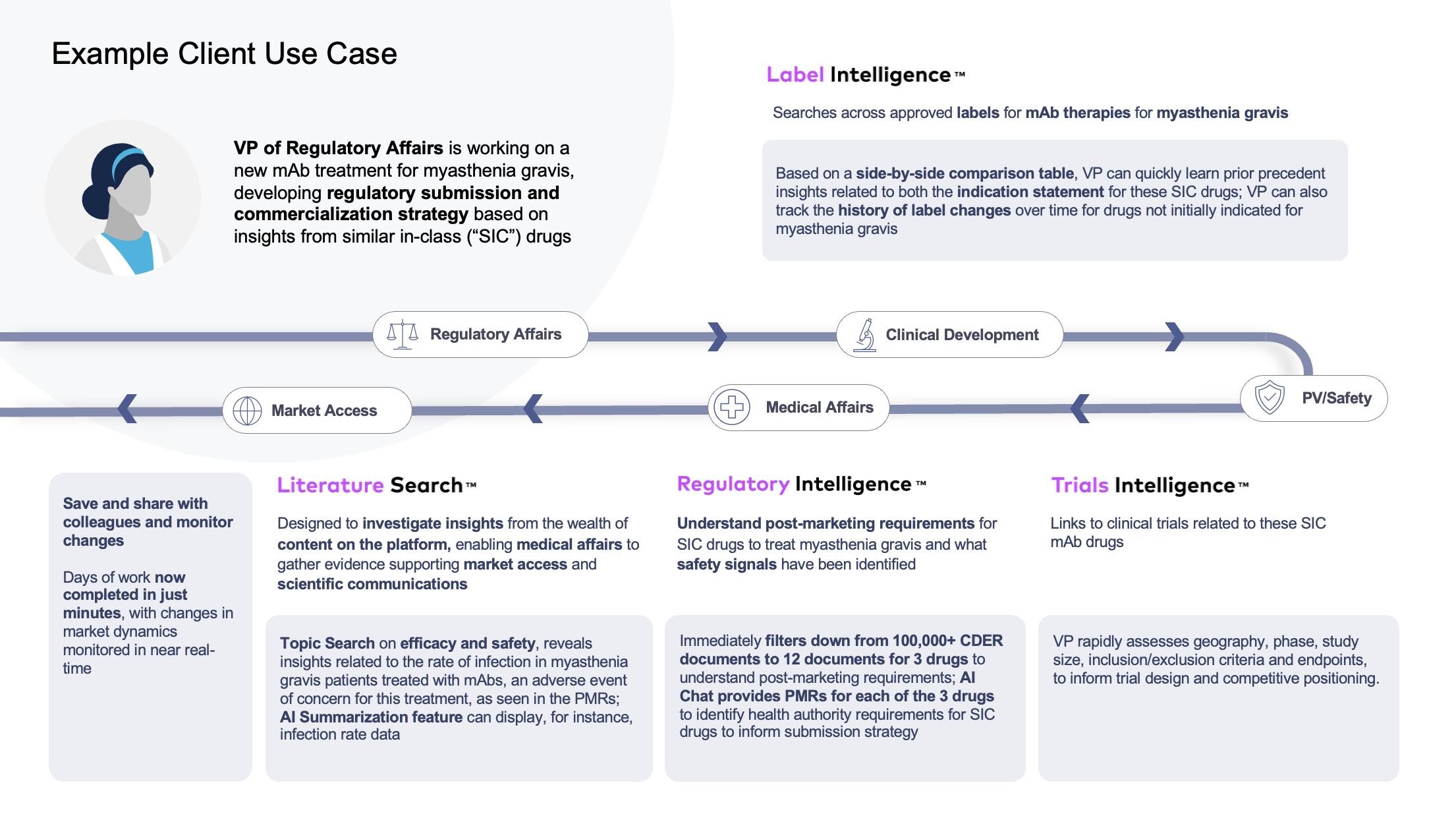
As the labeling and regulatory expert, you rely on a broad set of data sources, including not only labels but regulatory approval packages, clinical trial data, congresses, scientific literature and beyond. You educate, influence, and collaborate with peers across departments and geographies based on a comprehensive landscape view and an informed strategy. You anticipate and proactively prepare for health authority negotiations based on this intelligence.

Leverage Dr.Evidence as your Authoritative Source! Generate insights not only from labels, but from across the entire landscape of content.
De-risk while accelerating your global regulatory strategy in this dynamic market by leveraging all available prior precedent and competitive intelligence.
Proprietary large language models, trained exclusively on publicly available data curated by Dr.Evidence, power your workflows, summarizations and chat to drive efficiencies.

Regulatory Enforcement Actions
Missing critical label changes—new contraindications, safety warnings, or dosing modifications—can trigger regulatory citations, warning letters, and market withdrawal requirements, impacting product availability.
Commercial Impact
Delayed responses to label changes can disrupt supply chains. This can lead to lost market share and competitive disadvantage as rivals adapt faster to regulatory requirements.
Resource Drain
Your regulatory teams spend valuable time on routine monitoring instead of strategic initiatives that drive portfolio growth and accelerate approvals.
Your regulatory excellence shouldn’t depend on manual processes that scale poorly and fail predictably. With Dr.Evidence, you can track RLD labels in the platform and receive automatic email notifications when label content changes. No more risky manual monitoring. Spend more time on strategic initiatives that accelerate approvals and drive portfolio expansion, while working at your peak capabilities.
Upload draft labels in-platform and compare them section by section to on-market RLD labels to ensure your label content is accurate and complete.
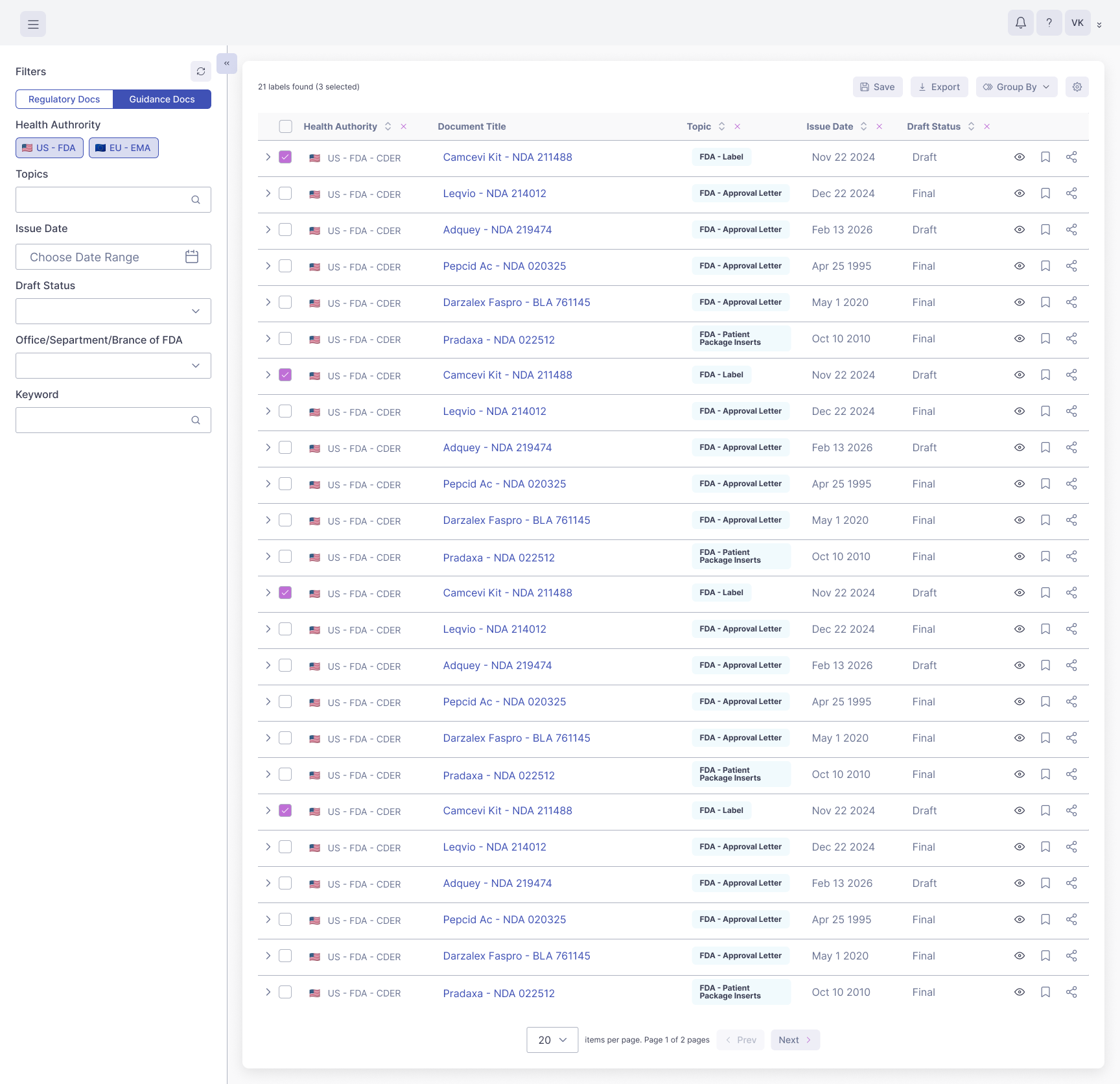
Effectively address skinny labeling opportunities with a comprehensive view of RLDs in the Dr.Evidence platform. Analyze relevant regulatory approval packages based on Orange book data, review patents for covered uses and indications, and identify the specific, narrow language you need in your label based on RLD labels.
Join the leading biopharma companies worldwide who've made Dr.Evidence their competitive advantage. Get the landscape intelligence platform that grows with you—from first submission to long-term commercial success.
Book a Demo